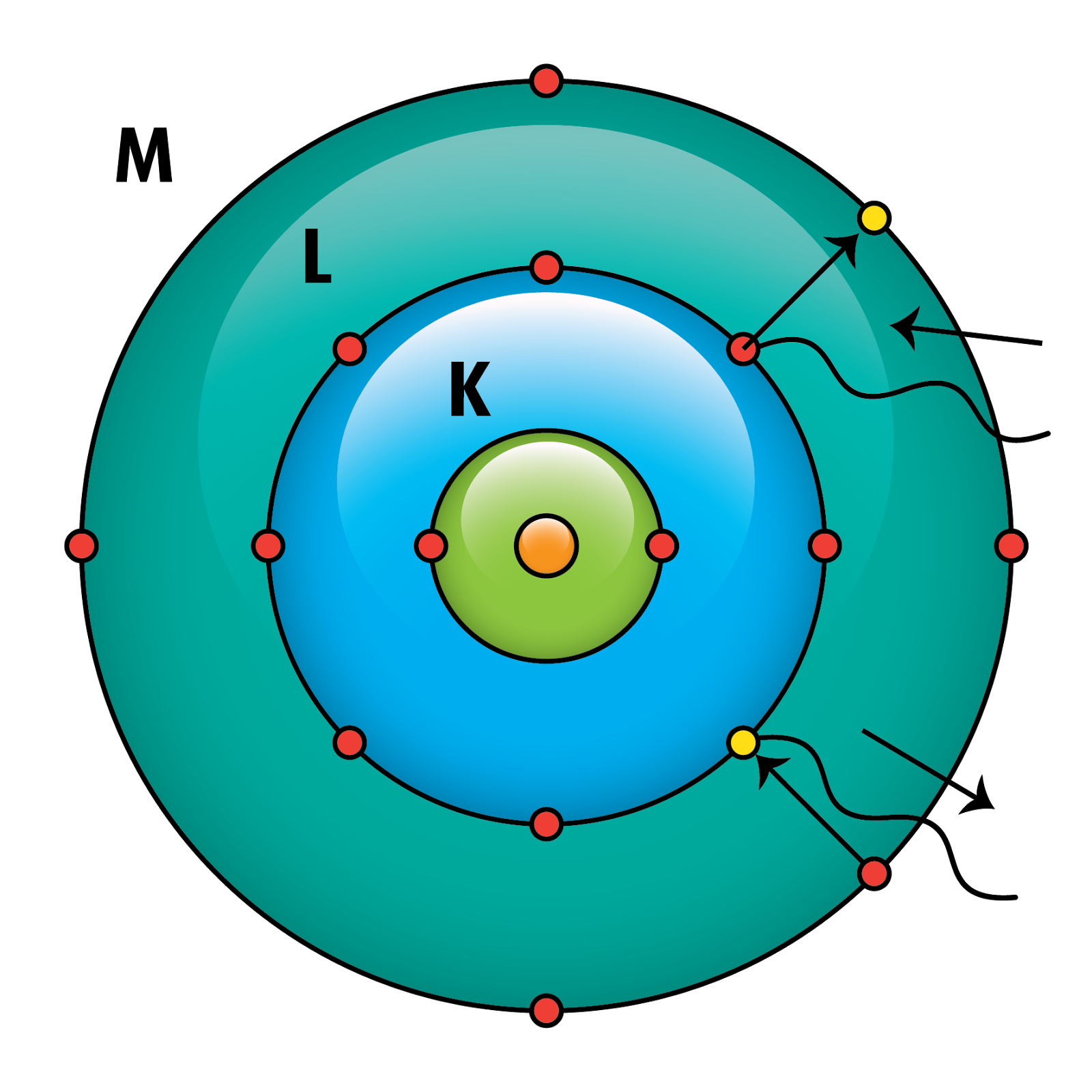
The popular theory of atomic structure at the time of Rutherford’s experiment was the plum pudding model. In the Bohr model of the atom, the nucleus contains the majority of the mass of the atom in its protons and neutrons. , 1997, Classical concepts in Bohr’s atomic theory (19131925), Physis: Riv. 
Using the Bohr model, determine the energy, in electron volts, of the photon produced when an electron in a. Bohr had adopted Ernest Rutherford’s model of the atom, according to which most of the mass of the atom is concentrated in a small central nucleus, while the electrons orbit the nucleus in planetary trajectories. It is the amount of energy that an electron gains when subjected to a potential of 1 volt 1 eV 1.602 ×1019 J 1 e V 1.602 × 10 19 J. Where \( \Re \) is the Rydberg constant, h is Planck’s constant, c is the speed of light, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. The gold foil experiment was conducted under the supervision of Ernest Rutherford at the University of Manchester in 1909 by Hans Geiger and Ernest Marsden. The electron volt (eV) is a convenient unit of energy for expressing atomic-scale energies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
